Studies have shown that hyperbaric oxygen therapy (HBOT) improves heart function mainly by reducing oxygen stress, regulating energy metabolism and inhibiting cell apoptosis.
The cardioprotective effects of HBOT are mainly evaluated by ELISA, fluorescent probes, transmission electron microscopy and reverse transcription- quantitative PCR (RT-qPCR).
As observed with the transmission electron microscope, using a 0.25MPa hyperbaric chamber for HBOT for 14 days (once a day) has improved mitochondrial morphology and reduced the number of autophagic vesicles.
HBOT significantly increased the opening levels of ATP, ADP, energy charge and mitochondrial permeability transition pores, but decreased the levels of AMP, cytochrome c and reactive oxygen species, which has been determined using RT qPCR or immunohistochemistry. In addition, HBOT significantly increased the gene or protein expression levels of eIF4E binding protein 1, mammalian target of rapamycin (mTOR), mitochondrial DNA, NADH dehydrogenase subunit 1, mitofusin 1 and mitofusin 2, while reduced the gene or protein expression levels of autophagy-related 5 (Atg5), cytochrome c, motility-related protein 1, and p53.
Data are expressed as the mean ± SD (n=5). *P<0.05; **P<0.01; ***P<0.001.
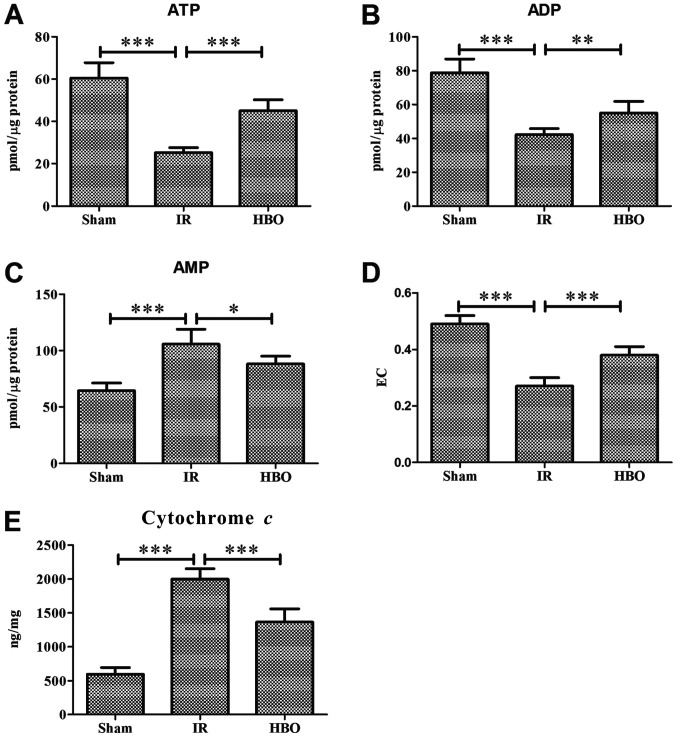
(ABD) ATP, ADP, EC are determined in Sham group, IR group and HBO group.
(CE) Use ELISA kit to detect in Sham group, IR group and HBO group
HBO, hyperbaric oxygen; EC, energy charge; IR, ischemia/reperfusion.
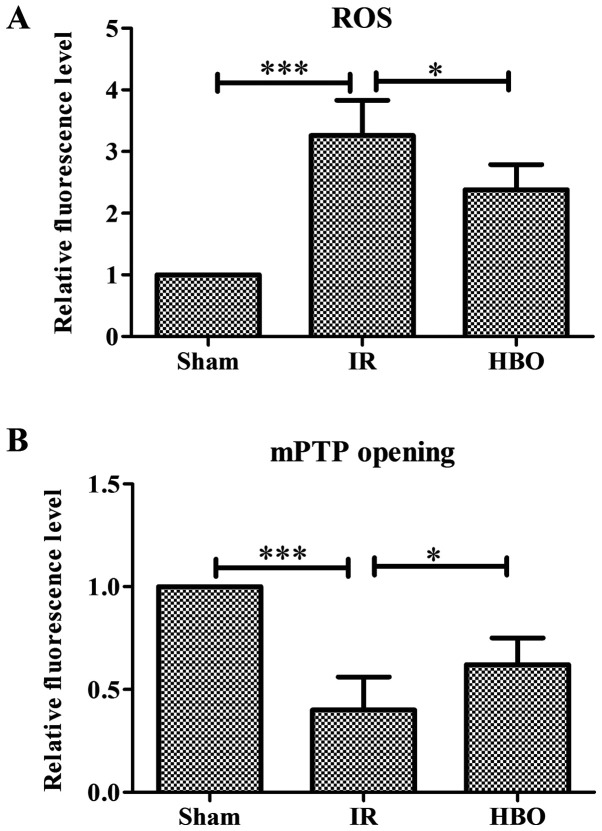
HBO, hyperbaric oxygen; ROS, reactive oxygen species; mPTP, mitochondrial permeability transition pore; IR, ischemia/reperfusion; DHE, dihydroethidium.
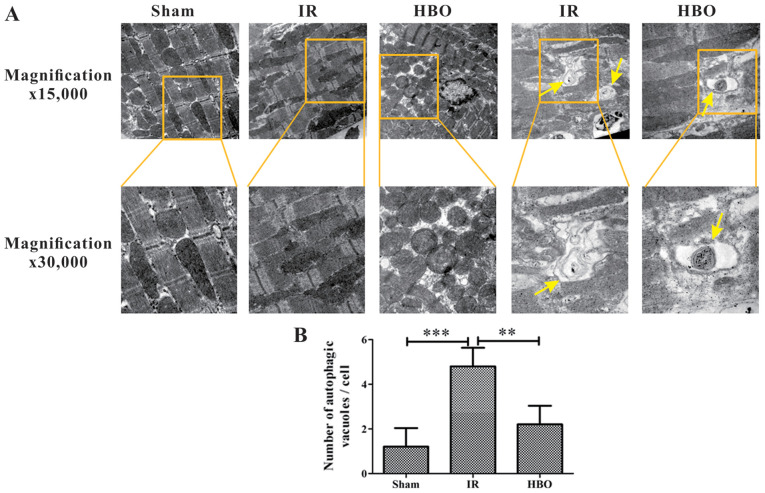
(A) Representative photomicrographs of myocardial tissue in the Sham group, IR group and HBO group. The mitochondria in the Sham group were normal, and no autophagic vesicles were observed. Compared with the Sham group, significant damage to mitochondria and an increase in the number of autophagic vacuoles were observed in the IR group. Compared with the IR group, only minor damage to mitochondria and a decrease in the number of autophagic vesicles (concentric circle changes) were observed in the HBO group. Yellow arrows indicate autophagic vesicles.
(B) Semi-quantification of the number of autophagic vesicles in Sham group, IR group and HBO group (n=5).
IR, ischemia/reperfusion; HBO, hyperbaric oxygen.
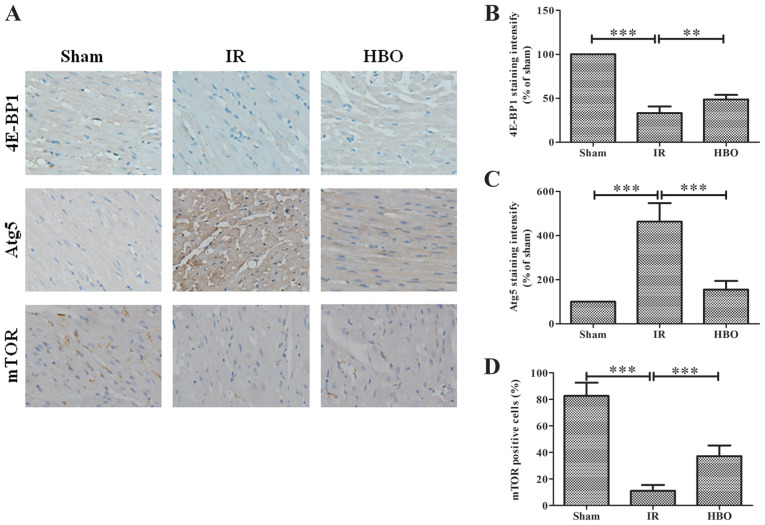
(A) Immunohistochemical staining of myocardial sections to detect the expression levels of 4E-BP1, Atg5 and mTOR in the Sham group, IR group and HBO group. The nucleus was stained blue, and the target protein was stained brown.
Semi-quantification of (B) 4E-BP1, (C) Atg5 and (D) mTOR expression levels in Sham group, IR group and HBO group. Data are normalized to the Sham group and expressed as percentages.
IR, ischemia/reperfusion; HBO, hyperbaric oxygen; Atg5, autophagy-related 5; 4E-BP1, eukaryotic initiation factor 4E binding protein 1; mTOR, mammalian target of rapamycin.
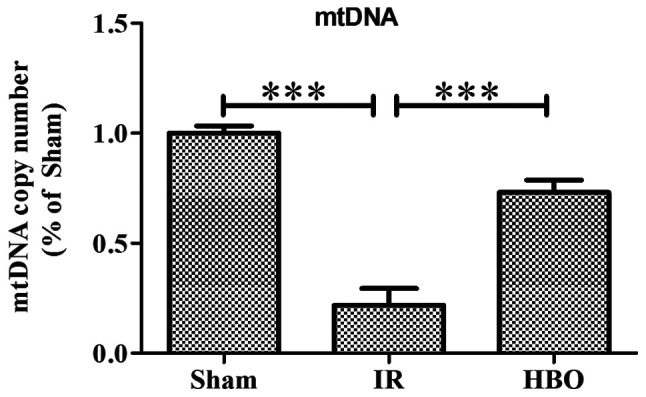
Data are normalized to the Sham group and expressed as percentages. mtDNA, mitochondrial DNA.
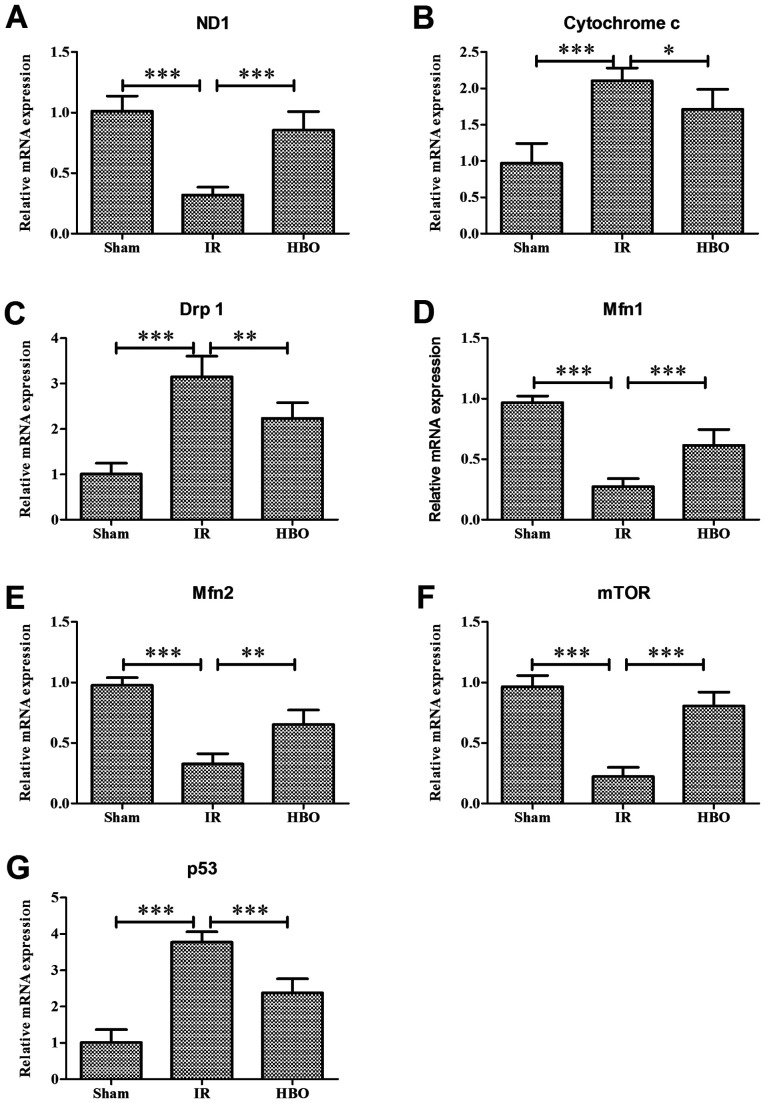
(AE) The effect of HBO pretreatment on the mRNA expression levels of mitochondrial dynamics related genes: (A) ND1, (B) cytochrome c, (C) Drp1, (D) Mfn1 and (E) Mfn2 in Sham group and IR group And the HBO group.
The effect of HBOT on the expression level of autophagy-related genes: (F) mTOR and (G) p53 were in the Sham group, IR and HBO group.
HBO, hyperbaric oxygen; IR, ischemia/reperfusion; ND1, NADH dehydrogenase subunit 1; Drp1, dynein related protein 1; Mfn1, mitofusin 1; Mfn2, mitofusin 2.
In summary, it has been observed that HBOT treatment protects cardiomyocytes during myocardial ischemia reperfusion injury (MIRI) by preventing mitochondrial dysfunction and inhibiting autophagy.
Therefore, these results provide new evidence to support the use of HBOT as a potential method of mitigating MIRI.
Cr: https://pubmed.ncbi.nlm.nih.gov/32901878/
Macy-pan has quality hyperbaric oxygen therapy chamber for sale, you can send a email to rachel@macy-pan.com or dial at 0086-180-1702-3916 if interested.

Discount Price, ship by DHL, door to door service, 5-7days delivery time, local technical support

We supply soft and hard chambers from 1.3-2.0ATA to reach different customers' needs

Create custom uniquea hyperbaric chambers to reach your target market

We have more than 50 distributors around world, looking forward to having you on board.