Hyperbaric oxygen therapy has been shown to improve human blood lipid metabolism and alleviate atherosclerosis. Studies have confirmed that it can prevent and combat thrombosis in ulcer and infection patients. The mechanism of hyperbaric oxygen in improving blood circulation, reducing blood viscosity and promoting thrombus dissolution and absorption is clear. It plays a role that cannot be replaced by drugs in nurturing cardiovascular health. Regular hyperbaric oxygen therapy is recommended for thrombosis prevention.
Sometimes in clinical practice, patients may ask "What is the effect of hyperbaric oxygen therapy on thrombosis in the human body?" This is a study that uses the whole blood thrombosis analysis system (T-TAS) to analyze the changes in thrombosis before and after hyperbaric oxygen therapy. The study proved that hyperbaric oxygen therapy can reduce the formation ability of human thrombus.
6 patients with skin and soft tissue ulcers and necrotizing fasciitis who received hyperbaric oxygen therapy.
The mean age was 65 years (range 54-75 years), with 4 males and 2 females.
The thrombosis formation ability was measured by T-TAS. T-TAS is a device that allows whole blood to flow through a simulated vessel wrapped in collagen and tissue factor, and can continuously measure the pressure curve from thrombosis formation to occlusion.
T-TAS allows the sample to flow through a uniform simulated vessel at a constant speed, making it easy to evaluate the blood's thrombosis formation ability. The flow rate of blood entering the simulated vessel will change according to the experimental conditions. Therefore, the thrombosis formation process is highly similar to the in vivo process and can be better evaluated than other devices.
2.0ATA, stable oxygen inhalation for 60 minutes, patients received 15 to 30 hyperbaric oxygen therapy sessions.
It was confirmed in all patients that hyperbaric oxygen therapy reduced thrombosis formation ability (as shown in the table below).
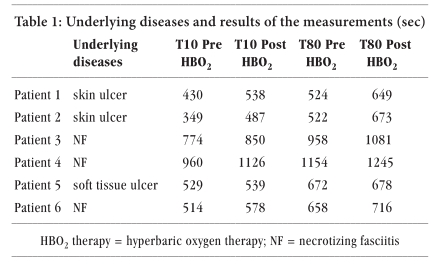
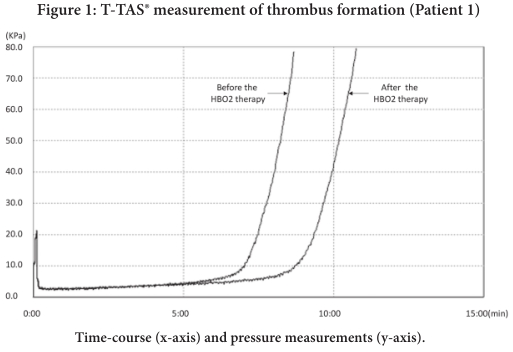
Before hyperbaric oxygen therapy, T10 (the start of white thrombosis formation) was 430 seconds, and T80 (complete microvascular occlusion due to thrombosis formation) was 520 seconds.
After hyperbaric oxygen therapy, T10 was extended to 538 seconds and T80 was extended to 649 seconds. This indicates that the adhesion ability of thrombus to the inner surface of the simulated vessel and the thrombosis formation ability have been reduced.
In this study, we determined that hyperbaric oxygen therapy can cause changes in thrombosis formation ability in ulcer or infection patients, which was confirmed by T-TAS simulating physiological conditions. To our knowledge, this is the world's first report using T-TAS. Our results are different from that of Monaca et al. who used MULTPLATE® and ROTEM®. This study supports the research of Peng et al. who used TEG® and Rotem. Our previous research corresponds to changes in platelet function in vitro high-pressure environments. Various studies have reported on the effect of hyperbaric oxygen therapy on thrombosis formation ability, many of which have concluded that coagulation ability is enhanced. One study suggested that hyperbaric oxygen therapy may increase thrombotic events.
However, in our report, in the simulated vessel close to the living state, hyperbaric oxygen therapy was found to reduce thrombosis formation ability. No report directly indicates that hyperbaric oxygen therapy increases thrombotic events in the body. Our current study confirms these findings.
In addition, our results suggest that hyperbaric oxygen therapy may improve wound healing. The changes in thrombosis formation ability after hyperbaric oxygen therapy may affect vascular improvement. In the human body, the thrombosis formation ability of blood depends on the platelet function and coagulation factor, the status of vascular endothelial cells forming blood passages, and mechanical factors such as flow rate and vascular contraction. These facets can further be enhanced through the production of high-quality hyperbaric oxygen chambers by specialized hyperbaric oxygen chamber manufacturers.
T-TAS has standardized simulated vessels and adjustable flow rate micro-pumps, so it is not affected by factors such as vascular characteristics or the number of vessels. Therefore, as our results indicate, hyperbaric oxygen therapy may affect blood flow by reducing thrombosis formation ability. As mentioned in our previous research, as a direct effect, pressurized dynamic stress may change platelet function.
By using hyperbaric oxygen therapy, we were able to detect changes in human thrombosis formation ability.
The measuring device we used can evaluate the thrombosis formation process under controlled blood flow, which has a significant advantage over the measuring devices used in previous reports. Therefore, unlike previous reports, the thrombosis formation ability showed a decrease. In the current study, we investigated patients with skin and soft tissue ulcers and necrotizing fasciitis. It is well known that hyperbaric oxygen therapy is effective for these diseases. Along with its effectiveness, the hyperbaric oxygen chamber price is also a crucial aspect that needs to be taken into consideration while evaluating the therapy's overall utility.
T-TAS is used to evaluate whether hyperbaric oxygen therapy is beneficial for improving coagulation function in other diseases such as sepsis and ischemic colitis. In this way, we may be able to apply hyperbaric oxygen therapy as a new treatment option for these diseases.
Conclusion: Hyperbaric oxygen therapy was found to reduce human thrombosis formation ability through T-TAS. We used T-TAS to evaluate the thrombosis formation process under simulated physiological conditions. We believe that T-TAS is a promising method for predicting the effects of hyperbaric oxygen therapy.
Macy-pan has quality hyperbaric oxygen chambers for sale, you can send a email to rachel@macy-pan.com or dial at 0086-180-1702-3916 if interested.

Discount Price, ship by DHL, door to door service, 5-7days delivery time, local technical support

We supply soft and hard chambers from 1.3-2.0ATA to reach different customers' needs

Create custom uniquea hyperbaric chambers to reach your target market

We have more than 50 distributors around world, looking forward to having you on board.